The $13B "Manual Tax"
Current centralized Quality Control infrastructure cannot support the velocity of decentralized, personalized medicines. We must move Quality Control inside the Manufacturing Suite.
The Failure Point
Patients wait. Therapies expire. Costs explode.

Hardware-Enabled SaaS
One-Time CapEx • GMP Consumables • Annual iPAT SaaS Licensing
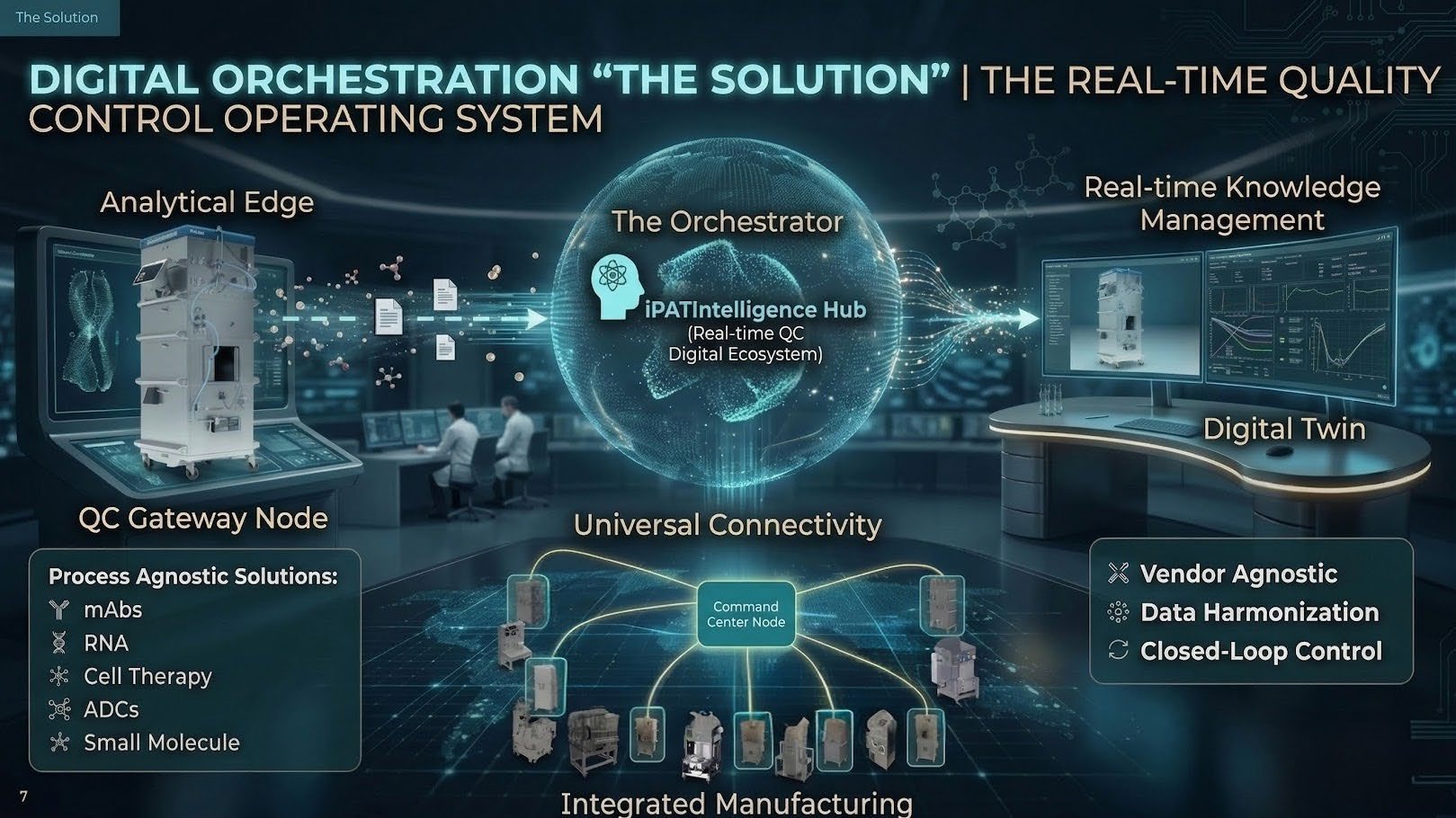
The Edge PAT Skid
"QC on Wheels". Mobile hardware bringing analytical insights into production. Enables simultaneous in-line multi-attribute analysis using Single-Use Flow Paths (GMP Requirement).
Learn MoreiPATintelligence Digital OS
A specialized MES and central data historian. Comes with pre-built regulatory filed/accepted models, meaning no PhD-level scientist is required on-site.
Learn MoreCMC Artificial Intelligence
Digital Twins that de-skill the lab. Predictive analytics for yield optimization and automated intelligence. Same analytical tools across the site, no tech transfer.
Learn MoreThe Regulatory Moat
Competitors sell hardware. We sell a Pre-Validated Regulatory Pathway.

$62M / 3-Year Contract
With FDA CBER to validate PAT tools as "Release Methods."
Type II DMF Strategy
Customers reference our DMF to treat the machine as a pre-approved "Drug Substance Engine."
76% CQA Coverage
Covered real-time. In-line results match offline analytics (<5% RSD).